• Osteotendinous junction (trauma/overuse or disease)
• Mid tendon (often occurs in degenerative tendons – tendinosis)
• Musculotendinous junction (vulnerable point, common site of tendinitis)
Tensile strength of a healthy tendon is normally more than twice the strength of its muscle and stronger than compact bone. A tendon with a cross sectional area of 1cm² is capable of supporting a weight of 500 – 1000kg (Jozsa and Kannus 1997). Therefore unless the tendon is unhealthy it will normally breakdown at the MTJ. The MTJ has a delicate ultrastructure and is often the weakest point of the muscle-tendon unit making it more susceptible to injury (Jozsa and Kannus 1997).
Tendon Injury Patterns
• Acute
• Chronic
Acute tendon strains
Excessive loads greater than 4 -10% of original length or direct contusion will cause acute injury to tendons. Under normal physiological circumstances tendons function in the toe and linear region of the stress-strain curve. Loads greater than this will cause damage most often at the MTJ. Muscles that cross two joints are predisposed – hamstring, rectus femoris, gastroc. This normally occurs when lengthening of the muscle combines with eccentric contraction.
Chronic/overuse tendon injury
Repeated loading has been shown to cause overuse tendon injuries when the destructive forces of applied stress exceed the reparative process. This occurs when the tendon is strained repeatedly to 4 – 8% of its original length until it is unable to endure further tension, where upon injury occurs (Hyman and Rodeo 2000).
This causes micro and macroscopic injury to collagen fibrils, noncollagenous matrix, and microvasculature causing inflammation. It is currently believed that the earliest pathophysiological changes occur in the paratendon (surrounding the tendon) causing paratendinitis. Continued microtrauma whilst in the inflammatory stage will lead to proliferation of synovial cells, fibroblasts and capillaries leading to fibrosis and thickening of the paratendon. Thus, tendonitis and paratendinitis are the earliest clinical manifestations of overuse tendon injury. If the damage progresses, tendinosis, partial tears and complete rupture may ensue (Jozsa and Kannus 1997).
Classification of Injury
Paratendinosis is inflammation of the paratendon only, either lined by synovium or not. It can be acute or chronic. Signs and symptoms: swelling, pain crepitus, local tenderness, warmth, and dysfunction.
Tendinitis is symptomatic degeneration of the tendon with vascular disruption and inflammatory repair response. Symptoms are inflammatory and proportional to the vascular disruption. It can be acute, subacute or chronic.
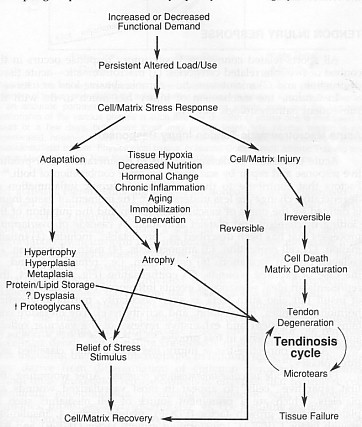
Tendinosis is when the tendon undergoes intratendinous degeneration due to cell atrophy, affecting its mechanical properties. The result of the pathological changes is decreased tensile strength that predisposes the tendon to injury. It is often asymptomatic since it is non inflammatory. It can be defined as “a change in tissue from a higher to a lower or less functionally active form”(Leadbetter 1992).
(Leadbetter 1992) illustrating Achilles paratendonitis +/- tendinosis
Classification of Tendon injuries and disorders
intratendinous degeneration due to atrophy (aging, microtrauma, vascular) can be nonsymptomaticsymptomatic degeneration of tendon with vascular disruption and inflammatory repair response
| NEW | OLD | DEFINITION |
| Paratendinitis | Tenosynovitis Tenovaginitis Peritendinitis |
inflammation of only the paratendon either lined by synovium or not. |
| Paratendinitis with tendinosis | Tendinitis | paratenon inflammation associated with intratendinosis degeneration |
| Tendinosis | Tendinitis | intratendinous degeneration due to atrophy (aging, microtrauma, vascular) can be nonsymptomatic |
| Tendinitis: | Tendon strain/tear acute, subacute chronic | symptomatic degeneration of tendon with vascular disruption and inflammatory repair response |
Adapted from (Leadbetter 1992)
Factors contributing to tendon injury (extrinsic/intrinsic)
• Excessive or Repeated loads – causes:
Poor biomechanics, joint laxity, leg length discrepancy – will alter direction, speed and size of force. e.g. foot hyperpronation has been linked to Achilles tendon injuries.
Muscle imbalances – weak muscle, tight muscle, poor timing of muscles can alter force distribution leading to strains.
Muscle fatigue, poor flexibility, – may increase load on tendons
Overweight – increased load
Training errors, poor equipment – e.g. prolonged exercise duration, could increase load
• Vascularity – if the tendon has a reduced blood supply (hypovascularity) it can lead to tissue hypoxia resulting in tendon cell atrophy contributing to tendon degeneration that results in tendinosis.
Hypovascularity has been linked most frequently to explain hypoxic degenerative tendon changes (O’Brien 1992). (Archambault et al 1995) reported that blood supply is irregular to the TA tendon mid-portion and at sites of twisting or around bony prominences. This study reported decreased vascularity in the TA tendon 4cm proximal to its calcaneal insertion. He suggested the cause was due to intermittent ischaemia during exercise – with subsequent reperfusion – leading to influx of oxygen derived free radical – these are highly reactive molecules that have a role in phagocytosis leading to tissue self destruction/degeneration.
Secondly, hypovascularity is linked to calcifying tendinopathy – persistent hypoxia may transform tenoblasts into chondroblasts that function anaerobically. Results in calcium deposition within the tendon.
Thirdly, regions of the tendon with poor vascularity will be less likely to cool over heated tissue caused by muscle activity, leading to exercise induced hyperthermia. This study reported that the intratendinous temperature of superficial digital flexor tendons of exercising horses (43 – 45º) exceeds the temperature at which fibroblast cell death occurs (42.5º). Thus, avascular regions of tendons may lack the ability to cool down the tissue leading to hyperthermic damage during exercise (Wilson and Goodship 1994).
• Genetic disorders/disease/infection – genetic collagen disorders like Ehlers-Danlos syndrome results in less cross-linking leading to weaker tendons. Rheumatoid diseases causes gross structural changes to tendons. Penetrating wounds can lead to tendon infections.
• Aging – results in stiffer, less compliant, weaker tendons with a reduced repair ability increasing their susceptibility to injury. This is due to decreased elastin, water and proteoglycan content, reduced collagen resynthesis and reduced vascularity and tenoblast activity (Hyman and Rodeo 2000).
• Endocrine factors – Diabetes Mellitus results in a compromise to tendinous microcirculation which may lead to tissue anoxia, cell necrosis thus predisposing a diabetic tendon to chronic paratendinitis or tendinosis (Jozsa and Kannus 1997). Hormonal changes in women can reduce the levels of estrogen, which has been linked to decreased collagen production.
• Nutritional deficiencies of cofactors (vitamin A, C and copper) important in collagen synthesis and cross-linking may affect tensile strength of tendons.
• Inactivity/Immobilization – (Jozsa and Kannus 1997) studied the effect of immobility on a human MTJ. Results showed the contact area between muscle cells and tendon collagen fibrils decreased to almost 50% in 3 weeks. There was also an increase in the amount of type III collagen at the MTJ, which is weaker than type 1 and a decrease in the amount of tenascin, an adhesive protein found in the MTJ. The overall effect will be to decrease tensile strength.
• Medication – corticosteroids (oral/injections) and certain antibiotics (flouroquinolone) can lead to pathology of the cell matrix. Excessive use of corticosteroid injections has been linked to tendon rupture (Jozsa and Kannus 1997). This may be due to its anti-inflammatory effect, as the injection reduces the pain, the patient will resume normal activity on the weak, degenerative tendon which could result in rupture.
• Skeletal maturity – osteotendonopathies like Osgood-Schlatters disease at the tibial tuberosity or Severe’s disease at the calcaneus can occur in the immature child due to tensile overload at the apophysis. Avulsions of small areas of the ossification center are found.